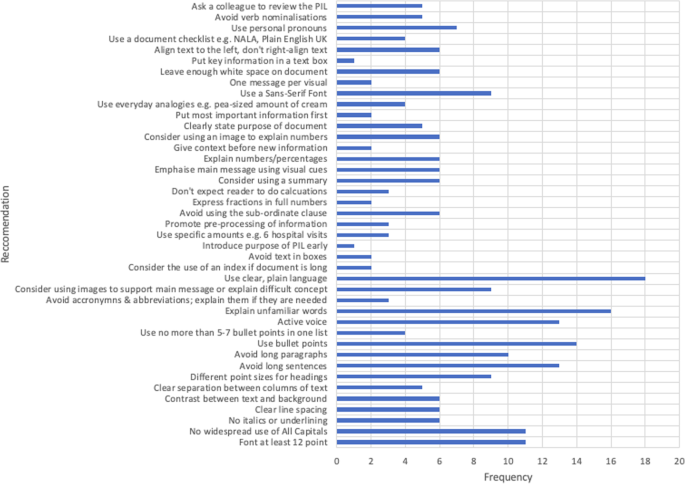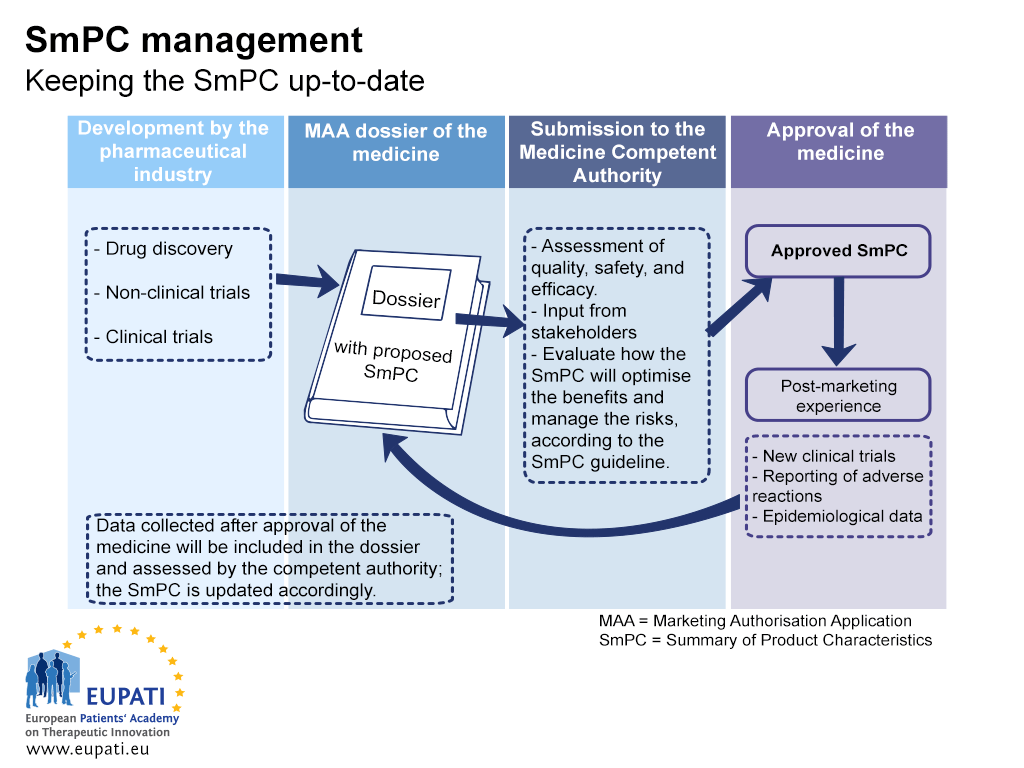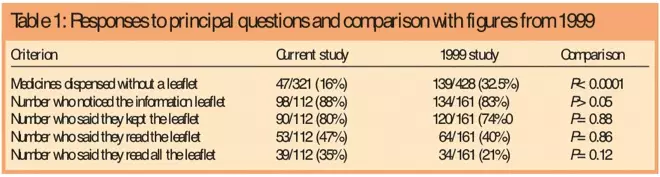
Patient information leaflets — impact of European regulations on safe and effective use of medicines - The Pharmaceutical Journal

Harmonization of malaria rapid diagnostic tests: best practices in labelling including instructions for use | SpringerLink

PDF) Testing of the readability of package leaflets as an initial step under the pharmaceutical care initiative towards increasing the safety of medicinal products

Preparing accessible and understandable clinical research participant information leaflets and consent forms: a set of guidelines from an expert consensus conference | Research Involvement and Engagement | Full Text

GUIDELINE ON THE READABILITY OF THE LABELLING AND LAYOUT OF THE INFORMATION ... 3.1 User testing ... The