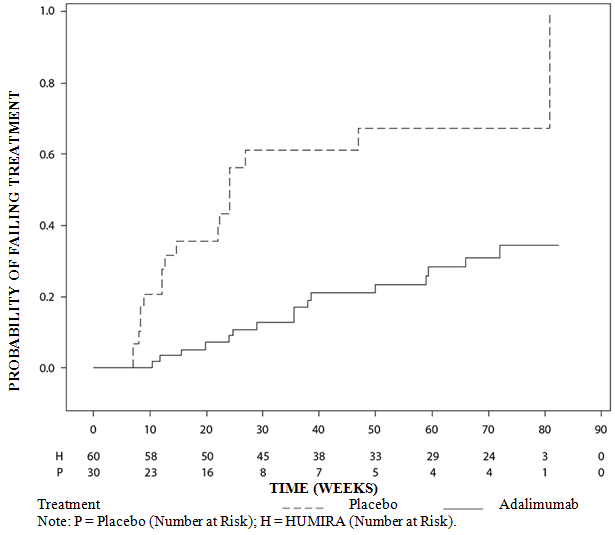
Long-term adalimumab efficacy in patients with moderate-to-severe hidradenitis suppurativa/acne inversa: 3-year results of a phase 3 open- label extension study - ScienceDirect
Humira 40 mg/0.4 ml Pre-filled Syringe and Pre-filled Pen (Great Britain) - Summary of Product Characteristics (SmPC) - (emc)